What is Hypoxia – Treatment For Hypoxia, Causes and Symptoms
Hypoxia is a condition in which the body or a region of the body is deprived of adequate oxygen supply at the tissue level,regular inhalation and exhalation can be a primary treatment for Hypoxia as adequate can be supplied to the blood.
Hypoxia may be classified as either generalized, affecting the whole body, or local, affecting a region of the body.
Although Hypoxia is often a pathological condition, variations in arterial oxygen concentrations can be part of the normal physiology, for example, during hypoventilation training or strenuous physical exercise.
Generalized Hypoxia occurs in healthy people when they ascend to high altitude, where it causes altitude sickness leading to potentially fatal complications: high altitude pulmonary edema (HAPE) and high altitude cerebral edema (HACE).
Hypoxia also occurs in healthy individuals when breathing mixtures of gases with a low oxygen content, e.g. while diving underwater especially when using closed-circuit rebreather systems that control the amount of oxygen in the supplied air.
Mild, non-damaging intermittent Hypoxia is used intentionally during altitude training to develop an athletic performance adaptation at both the systemic and cellular level.
Treatment for Hypoxia
To counter the effects of high-altitude diseases, the body must return arterial pO2 toward normal. Acclimatization, the means by which the body adapts to higher altitudes, only partially restores pO2 to standard levels.

technical sergeant staff in US-Air-Force treating Hypoxia
Hyperventilation, the body’s most common response to high-altitude conditions, increases alveolar pO2 by raising the depth and rate of breathing.
However, while pO2 does improve with hyperventilation, it does not return to normal.
Studies of miners and astronomers working at 3000 meters and above show improved alveolar pO2 with full acclimatization, yet the pO2 level remains equal to or even below the threshold for continuous oxygen therapy for patients with chronic obstructive pulmonary disease (COPD).
In addition, there are complications involved with acclimatization. Polycythemia, in which the body increases the number of red blood cells in circulation, thickens the blood, raising the danger that the heart can’t pump it.
In high-altitude conditions, only oxygen enrichment can counteract the effects of Hypoxia.
By increasing the concentration of oxygen in the air, the effects of lower barometric pressure are countered and the level of arterial pO2 is restored toward normal capacity.
A small amount of supplemental oxygen reduces the equivalent altitude in climate-controlled rooms. At 4000 m, raising the oxygen concentration level by 5 percent via an oxygen concentrator and an existing ventilation system provides an altitude equivalent of 3000 m, which is much more tolerable for the increasing number of low-landers who work in high altitude.
In a study of astronomers working in Chile at 5050 m, oxygen concentrators increased the level of oxygen concentration by almost 30 percent (that is, from 21 percent to 27 percent). This resulted in increased worker productivity, less fatigue, and improved sleep.
Oxygen concentrators are uniquely suited for this purpose. They require little maintenance and electricity, provide a constant source of oxygen, and eliminate the expensive, and often dangerous, task of transporting oxygen cylinders to remote areas.
Offices and housing already have climate-controlled rooms, in which temperature and humidity are kept at a constant level. Oxygen can be added to this system easily and relatively cheaply.
A prescription renewal for home oxygen following hospitalization requires an assessment of the patient for ongoing hypoxemia.
Causes
Oxygen passively diffuses in the lung alveoli according to a pressure gradient. Oxygen diffuses from the breathed air, mixed with water vapour, to arterial blood, where its partial pressure is around 100 mmHg (13.3 kPa).

In the blood, oxygen is bound to hemoglobin, a protein in red blood cells. The binding capacity of hemoglobin is influenced by the partial pressure of oxygen in the environment, as described in the oxygen–hemoglobin dissociation curve. A smaller amount of oxygen is transported in solution in the blood.
In peripheral tissues, oxygen again diffuses down a pressure gradient into cells and their mitochondria, where it is used to produce energy in conjunction with the breakdown of glucose, fats, and some amino acids.
Hypoxia can result from a failure at any stage in the delivery of oxygen to cells. This can include decreased partial pressures of oxygen, problems with diffusion of oxygen in the lungs, insufficient available hemoglobin, problems with blood flow to the end tissue, and problems with breathing rhythm.
Experimentally, oxygen diffusion becomes rate limiting (and lethal) when arterial oxygen partial pressure falls to 60 mmHg (5.3 kPa) or below.
Almost all the oxygen in the blood is bound to hemoglobin, so interfering with this carrier molecule limits oxygen delivery to the periphery. Hemoglobin increases the oxygen-carrying capacity of blood by about 40-fold,[21] with the ability of hemoglobin to carry oxygen influenced by the partial pressure of oxygen in the environment, a relationship described in the oxygen–hemoglobin dissociation curve. When the ability of hemoglobin to carry oxygen is interfered with, a hypoxic state can result.:997–999
Ischemia
Ischemia, meaning insufficient blood flow to a tissue, can also result in Hypoxia. This is called ‘Ischemic Hypoxia’. This can include an embolic event, a heart attack that decreases overall blood flow, or trauma to a tissue that results in damage. An example of insufficient blood flow causing local hypoxia is gangrene that occurs in diabetes.[23]
Diseases such as peripheral vascular disease can also result in local Hypoxia. For this reason, symptoms are worse when a limb is used. Pain may also be felt as a result of increased hydrogen ions leading to a decrease in blood pH (acidity) created as a result of anaerobic metabolism.
Hypoxemic Hypoxia
This refers specifically to hypoxic states where the arterial content of oxygen is insufficient. This can be caused by alterations in respiratory drive, such as in respiratory alkalosis, physiological or pathological shunting of blood, diseases interfering in lung function resulting in a ventilation-perfusion mismatch, such as a pulmonary embolus, or alterations in the partial pressure of oxygen in the environment or lung alveoli, such as may occur at altitude or when diving.
Carbon monoxide poisoning
Carbon monoxide competes with oxygen for binding sites on hemoglobin molecules. As carbon monoxide binds with hemoglobin hundreds of times tighter than oxygen, it can prevent the carriage of oxygen.Carbon monoxide poisoning can occur acutely, as with smoke intoxication, or over a period of time, as with cigarette smoking. Due to physiological processes, carbon monoxide is maintained at a resting level of 4–6 ppm. This is increased in urban areas (7–13 ppm) and in smokers (20–40 ppm).A carbon monoxide level of 40 ppm is equivalent to a reduction in hemoglobin levels of 10 g/L.The formula can be used to calculate the amount of carbon monoxide-bound hemoglobin. For example, at carbon monoxide level of 5 ppm, , or a loss of half a percent of their blood’s hemoglobin.
CO has a second toxic effect, namely removing the allosteric shift of the oxygen dissociation curve and shifting the foot of the curve to the left. In so doing, the hemoglobin is less likely to release its oxygens at the peripheral tissues.Certain abnormal hemoglobin variants also have higher than normal affinity for oxygen, and so are also poor at delivering oxygen to the periphery.
Altitude
Atmospheric pressure reduces with altitude and with it, the amount of oxygen.The reduction in the partial pressure of inspired oxygen at higher altitudes lowers the oxygen saturation of the blood, ultimately leading to Hypoxia.The clinical features of altitude sickness include: sleep problems, dizziness, headache and oedema.
Hypoxic breathing gases
The breathing gas in underwater diving may contain an insufficient partial pressure of oxygen, particularly in malfunction of rebreathers. Such situations may lead to unconsciousness without symptoms since carbon dioxide levels are normal and the human body senses pure Hypoxia poorly. Hypoxic breathing gases can be defined as mixtures with a lower oxygen fraction than air, though gases containing sufficient oxygen to reliably maintain consciousness at normal sea level atmospheric pressure may be described as normoxic even when slightly hypoxic. Hypoxic mixtures in this context are those which will not reliably maintain consciousness at sea level pressure. Gases with as little as 2% oxygen by volume in a helium diluent are used for deep diving operations. The ambient pressure at 190 msw is sufficient to provide a partial pressure of about 0.4 bar, which is suitable for saturation diving. As the divers are decompressed, the breathing gas must be oxygenated to maintain a breathable atmosphere.
Inert gas asphyxiation may be deliberate with use of a suicide bag. Accidental death has occurred in cases where concentrations of nitrogen in controlled atmospheres, or methane in mines, has not been detected or appreciated.
Other
Hemoglobin’s function can also be lost by chemically oxidizing its iron atom to its ferric form. This form of inactive hemoglobin is called methemoglobin and can be made by ingesting sodium nitrite as well as certain drugs and other chemicals.
Anemia
Hemoglobin plays a substantial role in carrying oxygen throughout the body,and when it is deficient, anemia can result, causing ‘anaemic Hypoxia’ if tissue perfusion is decreased. Iron deficiency is the most common cause of anemia. As iron is used in the synthesis of hemoglobin, less hemoglobin will be synthesised when there is less iron, due to insufficient intake, or poor absorption.:997–999
Anemia is typically a chronic process that is compensated over time by increased levels of red blood cells via upregulated erythropoetin. A chronic hypoxic state can result from a poorly compensated anaemia.
Histotoxic Hypoxia
Cyanide poisoning
Histotoxic Hypoxia results when the quantity of oxygen reaching the cells is normal, but the cells are unable to use the oxygen effectively as a result of disabled oxidative phosphorylation enzymes. This may occur in cyanide poisoning.
Symptoms
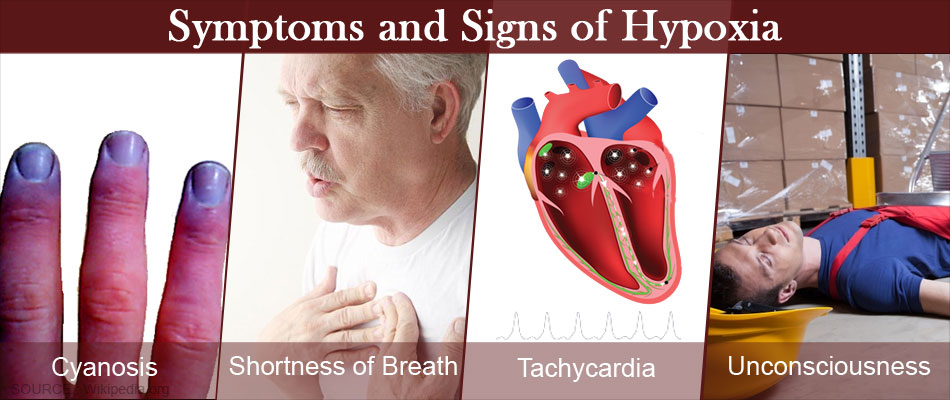
Although they can vary from person to person, the most common Hypoxia symptoms are:
- Changes in the color of your skin, ranging from blue to cherry red
- Confusion
- Cough
- Fast heart rate
- Rapid breathing
- Shortness of breath
- Slow heart rate
- Sweating
- Wheezing
Credit:
https://en.wikipedia.org/wiki/Hypoxia_(medical)#Cause





Leave an answer
You must login or register to add a new answer.