When hang-drying clothes, which is faster, indoors or outdoors
The answer to this question really depends on your situation. Let us state the question more concretely: we have a clean, wet load of laundry that just got washed. We want to dry the clothes as quickly as possible but only have the option of hanging up the clothes to dry. Would it be quicker to dry the clothes by hanging them indoors or outdoors? Before we can make sense of the answer to this question, we need to review the basics.
When a wet object dries, it is because the liquid water that is on the object evaporates to water vapor and diffuses out into the air. On the molecular scale, object wetness consists of many water molecules loosely and non-statically bonded to each other in liquid form, as well as bonded to the object. Drying consists of individual water molecules breaking free of their bonds to each other and to the object, and then flying out into the air. The speed at which a wet object dries is therefore determined by the net evaporation rate of the liquid water on the object.
In general, the drying rate of a particular wet object is influenced by three main factors: the temperature of the liquid water on the object, the concentration of the water molecles in the surrounding air, and the air flow rate.
1. Temperature.
As you may know, liquid water boils and forms water vapor when its temperature is high enough to break the intermolecular bonds holding the water molecules together. However, even when the water’s temperature is below the boiling point, some of the water molecules in the liquid break off into the air and become water vapor. How is this possible? The key is that not every water molecule in liquid water has the same energy. The temperature of a cup of liquid water does not tell us the exact energy of every single molecule. Rather, the temperature gives us an idea of the average energy of all of the molecules. Because of the random, jostling nature of thermal motion, some molecules in a cup of water end up with far more energy than the average, and some molecules end up with less energy than the average. The molecules with more energy than the average often have enough energy to break free of their liquid-state intermolecular bonds and shoot off into the freedom of air. In this way, some amount of water molecules will always evaporate from the surface of liquid water, no matter the temperature of the liquid water. The higher the temperature of the liquid water, the more molecules there are on the high end of the distribution with enough energy to evaporate. Therefore, the hotter an object, the quicker it dries, even if its temperature is below the boiling point of water.
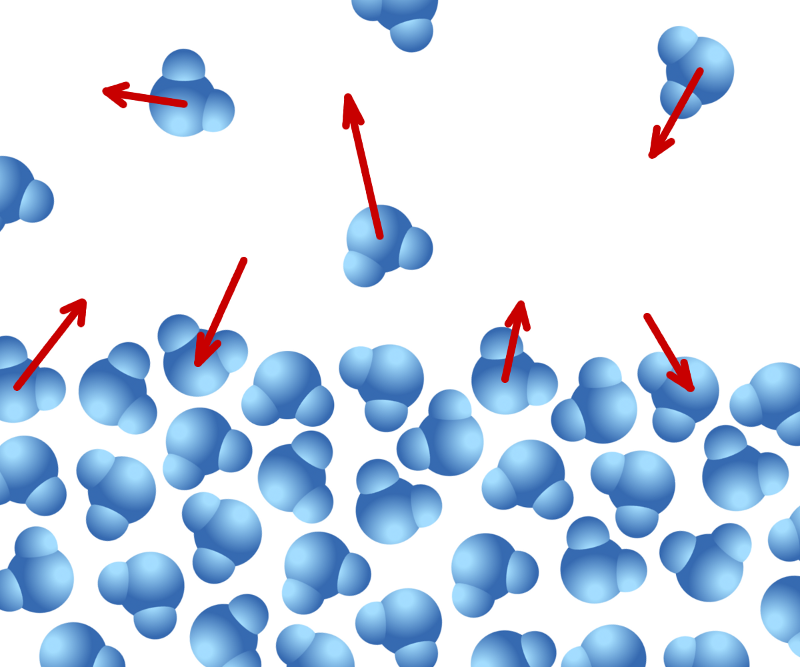
Water molecules on the surface of liquid water are evaporating into air at the same time that water molecules in the air are condensing onto the liquid water. In order for the liquid water to completely evaporate away, thus leaving the clothes dry, water molecules must leave the surface more quickly than they return. Public Domain Image, source: Christopher S. Baird.
Now let’s apply these concepts to our wet clothes. There are two common things which can warm up the hanging clothes and thus make them dry quicker: ambient hot air, and light. All else being equally, wet clothes hanging on a hot day will dry much quicker than wet clothes hanging on a cold day. Similarly, all else being equal, wet clothes hanging in the sunlight will dry much quicker than wet clothes hanging in the dark.
2. Concentration of Water Molecules in the Surrounding Air.
Now the story gets more complicated. Not only do water molecules leave the surface of the liquid water and shoot off into air, water molecules that are in the air also bump into the liquid water and stick there. If water molecules are joining the liquid water as quickly as they are leaving, the object will never dry. In fact, if water molecules join the liquid water faster than they leave, the object will get more wet over time and not less! In order to get an object to dry quickly, we want a lot of water molecules in the liquid water to break free and shoot off into the air, while at the same time, we want very few water molecules in the air to condense onto the liquid water. For fast drying, we therefore want the surrounding air to have a low concentration of water molecules. The concentration of water molecules in the air is commonly called the “humidity”. The more humid the surrounding air, the slower a wet object dries.
Returning to our wet clothes hung up to dry, they will dry more quickly in a room with dry air than in a room with humid air. If you live in a region of the world where the outdoor air is very humid (e.g. a tropical rain forest), it is likely that indoor air will be less humid than outdoor air. In this case, the clothes will generally dry more quickly indoors than outdoors. On the other hand, if you live in a region of the world where the outdoor air is very dry (e.g. a desert), it is likely that outdoor air is less humid and the clothes will dry more quickly outdoors. In most regions between these two extremes, the outdoor humidity varies significantly through the year, and even day to day.
Another thing to keep in mind is that if the room is small and not well ventilated, the humidity can build up in the room as the hanging clothes dry. The water molecules coming off the clothes as they dry will fill up the room’s air more and more, giving more chance for them to land back on the clothes. In this way, wet clothes in a small, unventilated room will dry more slowly than wet clothes in a large, well-ventilated room, all else being equal. If the outdoor air has low humidity, “effective ventilation” can simply mean opening a window.
3. Air Flow Rate.
If the air is perfectly motionless, the water vapor that comes off of an object that is drying will tend to stay mostly near the surface of the object. Although the water molecules in the air will diffuse throughout the whole room because of their random jostling motion, diffusion is a slow process. Because the evaporated water molecules mostly stay close to the surface of the object, they have a lot of opportunity to bump into the object, condense back to liquid, and slow down the drying process. In contrast, if the air is moving, the water molecules that evaporate are swept away before they have a chance to reattach to the object. In this way, air flowing past the surface of a wet object speeds up evaporation. More accurately, air flow will only speed up evaporation if the new air flowing past the object has a lower humidly than the air it is displacing. The harder we blow dry air across an object, the faster it will dry. Applying this concept to our wet clothes hung up to dry, they will indeed dry faster in the breeze than in a spot with no wind, all else being equal.
With these three factors in mind, clothes will dry the fastest when hung up outdoors in a dry, sunny, hot, windy climate. Note that hanging clothes in direct sunlight will cause the colors in the clothes to fade. For those who live in a humid, overcast, cold, non-windy climate, clothes will dry the fastest when hung indoors in a room with low humidity, high temperature, lots of light, and moving air. In other words, turn up the heat, turn up the dehumidifier, turn on some fans, and open the curtains (but not the windows) if you live in the wrong kind of climate and want to maximize drying rates. For those of us who live in regions that are between these two extremes, to determine whether outdoors or indoors is better, we need to ask ourselves the questions: Is the outdoor air less humid than the indoor air? Is the outdoor air warmer than the indoor air? Is outdoors brighter than indoors? Is outdoors breezier than indoors? If the answer to most of these questions is “yes”, then the clothes will dry faster outdoors.
The worst place to dry clothes is in a damp, dark, cold, unventilated basement – that is, unless you place the clothes in a clothes dryer machine that sits in the basement – then it’s the best place to dry clothes.
Credit:https://wtamu.edu/~cbaird/sq/2015/03/06/when-hang-drying-clothes-which-is-faster-indoors-or-outdoors/


Leave an answer
You must login or register to add a new answer.