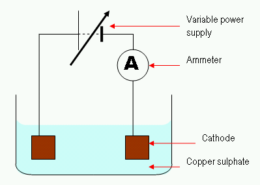
Why is Copper Discharged in the Electrolysis of Copper (II) Sulphate Solution?
In the electrolysis of copper (II) sulphate solution, copper ions are discharged in proportion to the amount of copper (II) ions dissolved in the solution. The discharged ions are removed from the solution by a variety of processes, including absorption by solid matter or dissolving in water.
In copper (II) sulphate solution, the copper ions are bound to anions (sulphate) in a redox reaction. The overall reaction is:
2Cu(s) + 3H2O (l) -> 2Cu2+ (aq) + 2SO4(aq)
The copper ions are released as a result of the reduction of the SO4 ions, which results in the formation of hydrogen gas.
The discharge of copper in the electrolysis of copper (II) sulphate solution is due to the following reasons:
1. The dissolution of CuSO 4 in water liberates Cu2+ ions. These ions combine with electrons from water molecules to form H2O2. This second reaction is exothermic and releases heat, which causes the discharge of copper.
2. The dissociation of CuSO 4 into Cu and SO 4 2- ions also releases Cu2+ and SO4+. These ions combine with electrons from water molecules to form H2O and HSO4-. The net result is an increase in the concentration of these salts and a decrease in the concentration of CuSO 4 .


Leave an answer
You must login or register to add a new answer.